
Nail lesions
The most common nail lesion is the crack, perpendicular to the ground. These cracks are often the result of overpressure on the nail tip, when the nail is longer than the sole. In that situation, each step the elephant makes results in heavy forces on the nail tip during the phase when the elephant unwinds its foot. As demonstrated clearly in the slow-motion video below, the tip of the elephant's nails hardly touches the ground.


When the nail becomes too long like in the Asian elephant nail in this photo, the excessive pressure on the nail edge can cause a small crack that enlarges over time if not properly treated.

Cracks in the nails of an African elephant
When left untreated, these cracks will become larger and can affect deeper structures, resulting in an abscess, pododermatitis or even osteomyelitis of the phalangeal bones. If only the horn-producing tissue is involved, we usually call this a pododermatitis. When the infection is trapped in the underlying tissue, an abscess can easily develop.
Onychia is an infection or trauma to the horn lamellae of the nail, which may result in complete loss of the nail.

Pododermatitis in the nail of an African elephant. Note the excessive wear of the sole.

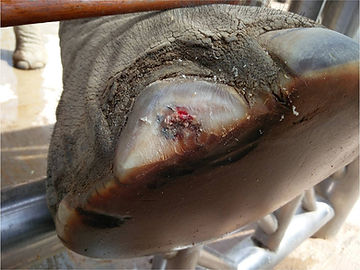
Nail abscess with osteolysis in an Asian elephant.

Nail abscess with complete loss of the 2 distal phalanges osteolysis in an Asian elephant.
If phalanges are affected as the result of a deep nail abscess and pododermatitis, the recommended treatment of such an osteitis is the surgical removal of the affected bones. Click here to read more about this treatment.
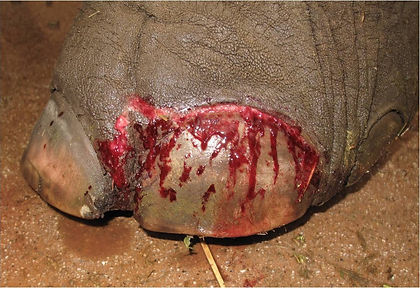
Traumatic onychia in an Asian elephant.

Complete nail loss due to traumatic onychia in an Asian elephant.
Diagnostic procedures nail lesions
Use your eyes: check if there is any visible lameness. Describe the visible lesions.
Use your hand: does the affected area feels warm? Is it painful when pressure is applied?
Use your nose: if there is a wound, try to identify the smell of necrosis. Take a swab for bacterial culture.
Additional diagnostic steps:
If there is a deep lesion: radiographs should be taken to look at the integrety of the underlying bony structures (phalanges and phalangeal joints).
Thermography may help to identify if the affected area has a higher temperature than the surrounding tissues.
Radiography of the elephant's foot
A powerful portable X-ray machine (100 kV or more) is required to visualize the bony structures in the elephant foot. Digital plates largely increase the quality of the image. It is important to work under safe conditions. Under free contact management, the fore foot can be positioned on a stand for the oblique palmar-dorsal shooting direction. The angle required for visualization of the phalangeal joints is indicated in the images below.

Oblique palmar-dorsal image of the hind foot under free contact management condition (Mumby et al 2015)

Positioning for the oblique dorsal-plantar image of the hind foot, this foot can be can be positioned on a stand (Mumby et al 2015).
A different approach is required when working under protected contact management. The elephant needs to be trained to position its legs on a horizontal bar of the training wall. It should also be accustomed to the proximity of the X-ray-machine, the plate and the protective clothing of the operators.

Lateral shooting position of the right front foot of an Asian elephant under protected contact. The angle of the beam depends on which phalanges need to be visualized. To avoid superposition and depending on the selected phalanges, the beam should have a more or less oblique direction. (Courtesy Rotterdam Zoo).

Oblique dorsal-plantar shooting position of the left rear foot (Courtesy Rotterdam Zoo). Click here to view the radiograph.

Oblique palmar-dorsal shooting postion of the right front foot of an Asian elephant under protected contact. Note that the X-ray machine is positioned upside down (use a support block to protect the electric wires)!
(Courtesy Rotterdam Zoo and Emmen Zoo). Click here to view the radiograph.

Lateral shooting postion of the left hind foot of an Asian elephant under protected contact (Courtesy Rotterdam Zoo). Click here to view the radiograph.
Some examples of radiographs of the distal part of the right front leg of a 24 yr-old female Asian elephant at Rotterdam Zoo (the Netherlands) are shown below. The elephant is under standing sedation for an unrelated reason.

Lateral shooting position of the right front leg (distal part of the radius and ulna, Courtesy Rotterdam Zoo). Click here to view the radiograph.

Anterior-posterior shooting position of the right radio-carpal and ulnar carpal joint AP front leg (Courtesy Rotterdam Zoo). Click here to view the radiograph.

Posterior-anterior shooting position of the right radial-carpal and ulnar-carpal joint (Courtesy Rotterdam Zoo). Click here to view the radiograph.

Lateral position of the right carpal joint (Courtesy Rotterdam Zoo). Click here to view the radiograph.

Lateral shooting position of the right foot (Courtesy Rotterdam Zoo). Click here to view the radiograph.

Lateral shooting position of the right tarsal joint (Courtesy Rotterdam Zoo). Click here to view the radiograph.

Lateral shooting position of the right tarsal joint (Courtesy Rotterdam Zoo). Click here to view the radiograph.

Posterior-anterior shooting position of the right tarsal joint (Courtesy Rotterdam Zoo).
Stereo radiography
A technique, which is called stereo radiography can help interpreting the X-rays. Two radiographs of the same areas are made each of them taken 10 cm more lateral from the other. Digital images were converted to bitmap (BMP) format to preserve image quality. The 3D
stereoradiograph images can be constructed using special software (Bentley 2021). The images
can be viewed with red-cyan 3D glasses.

Illustration of how 3D stereoradiograph images are produced. (a) Original radiograph that provides the ‘‘left’’ image for the 3D Anaglyph software. (b) Paired radiograph used for ‘‘right’’ image. (c) Completed stereoradiograph that has been inverted with red-cyan settings applied. (Bentley 2021)
Thermography of nail lesions
Thermographic imaging can be used to measure the absolute temperature and the difference in temperature between the lesion and its surrounding tissues. Below a severe case of a nail abscess-related osteomyelitis and osteolysis of Ph4 in an old Asian elephant bulls is shown. The thermografic image shows a low temperature of the skin that covers the affected are. This is suggestive for a large necro-purulent process.



The thermographic image shown here demonstrates the low temperature of the skin covering the abscess, associated with deep lesions of D4 of the left front leg of an Asian elephant bull, involving a lot of necrosis and complete loss of the distal phalanx of the toe. On radiology this bone is completely missing and the distal part of the toe shows an irregular surface: Osteomyelitis and purulent arthritis of the Ph3-4 joint of D4.
Treatment of nail lesions
Pedicure is usually the treatment of choice in cases of nail lesions.
There are a few principles to be respected in pedicure:
-
Use proper equipment
-
Always remove all abnormal horn tissue (undermined, infected) and loose nail flaps.
-
Make the transition from healthy horn to the deepest point of the lesion as smooth as possible. Directly adjacent to the lesion, the horn must be flexible and as thin as a piece of paper. Permanently check this flexibility by gently pressing the area where just cut away the horn.



The most important pieces of equipment needed for pedicure are 2 hoof knives (left and right handed), a scalpel and a sharpening stone for hoof knives, preferably with round edges (see photos above). Just with hoof knives alone, most of the clinical pedicure treatment can be done. A scalpel can be useful when very small pieces of horn are to be removed adjacent to a lesion.
A horse hoof rasp is a useful tool to shorten the nail and remove excessive horn from the sole. Care should be taken NOT to make the nail edges round, what is often practiced (see below).
A strong nail brush is needed for cleaning the nail prior to pedicure and a smaller brush can be used to remove dirt from areas that cannot be reached by a large brush.
Small wood carving knives can be used when thin layers of nail horn are to be removed (similar to the use of a scalpel).

Pedicure equipment

Very often the tip of the nails are made round by rasping the corners. There is no justification for doing so rather than a cosmetic one. However, one should avoid to remove tissue from the nail wall/nale sole junction (comparible to the "white line" in the horse hoof) as this junction is a very important barrier against infiltration of dirt and pathogens.
So the advice is: do not file the nail corners to leave the entire white line in tact.



Situation before a pedicure session of a deep nail crack


Situation after a pedicure session of a deep nail crack
Pedicure around a (deep) crack serves 2 main purposes:
-
Draining of infected area
-
Removing pressure on the wound, which enables the regeneration of destroyed horn lamellae.
In order to achieve both goals, the deepest point of the crack has to be freed from covering horn. One should start making the horn wall thinner at a distance of several centimeters from the lesion and continue the pedicure towards the deepest point in a gradual way. Each nail crack should be considered as being a wound, because the crack has usually damaged the horn lamellae. When the pedicure has reached the wound area, it is of utmost importance that the edges bordering the wound are made as thin and flexible as possible. Check this by pressing your finger on the horn adjacent to the wound.
Large cracks usually need to be treated in several sessions. Bleeding may occur when the lamellae are cut, which is no direct reason to worry! Pain reactions of the elephant will tell the operator when the session should be stopped and continued a few days later
Cuticle lesions
The cuticles form a natural barrier against dirt and pathogens. When they overgrow the nails, this protective barrier is weakened and infiltration of microoganisms may result in lesions in the horn lamellae underneath the nail (onychia). Sweat glands are embedded in the cuticles.
Overgrown cuticles may become hard and crack or form interdigital callus. When abnormal horn tissue blocks the sweat glands, fluid pockets may be formed. This can be a painful process and needs to be treated.

Minor cuticle lesion in a African elephant, which may be connected to a deeper lesion underneath the nail. Explorative pedicure is probably needed to find the cause (Courtesy: Barcelona Zoo)

Extensive overgrown cuticles with feathering and interdigital callus formation (Fowler & Mikota 2006)

Interdigital callus formation in an Asian elephant (Courtesy: Susan Mikota)

Sweat glands are present in large numbers in the cuticles. When the cuticles are overgrown, they may form pockets in which the sweat-fluid may accumulate ("blisters"or "blebs"). When cutting in such a fluid-filled pocket, the contents may squirt out (see video; courtesy: Susan Mikota).

Careful (!) trimming of the cuticles is only indicated when they have overgrown the nail (Courtesy: Susan Mikota)

Applying oil on the cuticles will make them softer and may resolve the problem of cuticle overgrowth in most situations.
Example of a pedicure procedure of a pododermatitis in conjunction with a cuticle lesion in an African elephant (courtesy Barcelona Zoo)

Pododermatitis in the nail of an African elephant. Note the excessive wear of the sole and the large defect of the cuticle.

Second step: follow-up the necrotic tissue and find the connection with the cuticle defect. Note that a large part of the nail was undermined, causing the cuticle defect.
First step in pedicure: making the horn on each side of the lesion thinner and removing necrotic tissues.

By frequently removing all necrotic tissues and keeping the edges of the wound thin, the horn lamellae can produce healthy horn again.
SUCCESSFUL TREATMENT OF DIGITAL OSTEITIS BY INTRAVENOUS REGIONAL PERFUSION OF CEFTIOFUR IN AN AFRICAN ELEPHANT (Loxodonta africana)
(Dutton C.J., Delnatte P.G., Hollamby S.R., and Crawshaw G.J. Journal of Zoo and Wildlife Medicine 48(2): 554–558, 2017)
A 41-yr-old African elephant (Loxodonta africana) presented with a swollen third digit of the left
forelimb and a 2-cm hole in the pad. Corrective trimming, topical treatments, and an oral antibiotic resulted in apparent resolution; however, it reoccurred after 4 mo. Radiographs suggested bone lysis in the third phalanx, with the primary differential diagnosis being septic osteitis. Flushing with metronidazole solution and intravenous regional perfusion (IVRP) of the foot were commenced. A tourniquet was applied just above the carpus, an interdigital vein was identified by ultrasound, and into this vein 2 g (20 ml) of ceftiofur sodium solution, followed by 60 ml of heparinized saline, was administered. The foot was kept raised for 25 min and then the tourniquet was removed. IVRP was repeated every other day for 70 treatments over 6 mo. Healing occurred, which was confirmed radiographically. IVRP offers an excellent treatment modality in a well-trained elephant.
SURGICAL REMOVAL OF INFECTED PHALANGES FROM AN ASIAN ELEPHANT (Elephas maximus)
Gage, L. Blasko D, Fowler M.E. and Pascoe J.
Joint Conference AAZ/WDA/AAWV
After unsuccessful antimicrobial treatment of an osteitis in a 40 yr-old Asian elephant, surgical removal of the affected phalanges resulted in the complete healing of the foot. Click here to read the report.
Literature
-
Bentley C.E., Cracknell J.N., Kitchener A.C., Pereira Y.M., Pizzi R. 2021. Improved diagnosis of foot osteoarthritis in elephants (Elephas maximus, Loxodonta africana) using stereoradiography. Journal of Zoo and Wildlife Medicine 52(1): 67–74, 2021.
-
Dutton C.J., Delnatte P.G., Hollamby S.R., and Crawshaw G.J. 2017. Successfull treatment of digital osteitis by intravenous regional prefusion of ceftiofur in an african elephant (Loxodonta africana). Journal of Zoo and Wildlife Medicine 48(2): 554–558, 2017).
-
Gage, L. Blasko D, Fowler M.E. and Pascoe J. 1995. Surgical removal of infected phalanges from an asian elephant (Elephas maximus). Joint conference AAZV / WDA/ AAWV.
-
Mumby, C., Bouts, T., Sambrook, L., Danika, S., Rees, E., Parry, A., Rendle, M., Masters, N. and Weller, R. (2013), Validation of a new radiographic protocol for Asian elephant feet and description of their radiographic anatomy. Veterinary Record, 173: 318-318. https://doi.org/10.1136/vr.101696.
-
Fowler M.E. and Mikota S.K. 2006. Biology, Medicine, and Surgery of Elephants. 271-290.


